慈濟大學研究團隊提出出血性登革熱雙重打擊假說
Research team of Tzu Chi University reports the two-hit hypothesis on the elicitation of dengue hemorrhage fever
As the dengue epidemic continues to spread in Taiwan, with hundreds of people died of severe dengue infections, the research team of Tzu Chi University has achieved a major progress on the establishment of a two-hit model of the dengue hemorrhage fever (DHF).
Primary dengue virus infection can induce mild symptoms, which is known as dengue fever, a self limited disease. However, instead of inducing protective immunity, secondary infections of dengue virus may induce life threatening DHF. Therefore, abnormal immune responses were though as part of the pathophysiology of DHF. Probably because of the pathogenic mechanism of DHF remains unclear, no specific treatments and effective vaccines are currently available.
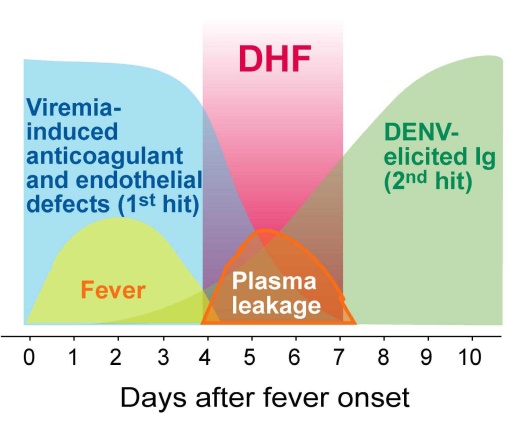
An integrated group of three research teams of Professor Hsin-Hou Chang and Der-Shan Sun at Department of Molecular Biology and Human Genetics, Tzu-Chi University, and Professor Wen-Sheng Wu at Department of Laboratory Medicine and Biotechnology, College of Medicine, Tzu Chi University, has proposed a two-hit hypothesis for dengue hemorrhage fever: viremia of dengue virus is the first hit, and dengue-elicited autoantibody is the second hit. When damages that induced by both hits over the pathological thresholds, the hemorrhage pathogenesis is elicited. Using this hypothesis, the research group of Tzu-Chi University has successfully demonstrated that concurrent dengue virus and autoantibodies treatments are able to elicit hemorrhage pathogenesis in a mouse model. The relevant studies have been published in the international journals “Thrombosis and Haemostasis” and “Journal of Immunology” 1,2, and the 10th Asia-Pacific Medical International conference 3.
Professor Chang has suggested that if the two-hit model is indeed involved in the hemorrhage pathogenesis, screening of autoantibody titers or other pathogenic factors may be applied prior to the elicitation of hemorrhage pathogenesis. With such surveillance, DHF might be treated at an early stage. In addition, as the dengue nonstructural protein NS1 can elicit autoantibody in the host, how to prevent the elicitation of autoantibody is one of the major challenges to develop a safe and effective dengue vaccine.
Figure. The two-hit hypothetical model; adopted from reference 2.
在全台登革熱疫情持續延燒之際,驚聞有上百人死於重症之出血性登革熱。慈濟大學研究團隊不落人後,在出血性登革熱的病理機轉研究上有新的進展。
通常第一次感染登革病毒可誘發症狀輕微之登革熱,但是二次感染登革病毒有可能誘發出血症狀,甚至導致重症及死亡,造成所謂之出血性登革熱。由於二次感染登革病毒才能誘發出血性登革熱,同樣於二次感染中會被喚醒的免疫反應,被認為與出血熱及重症的發生有關。但是因為致病機轉並不清楚,目前出血性登革熱並沒有特效藥及有效疫苗,所以只能以改善環境衛生,及支持性療法,實施消極而被動的防治。
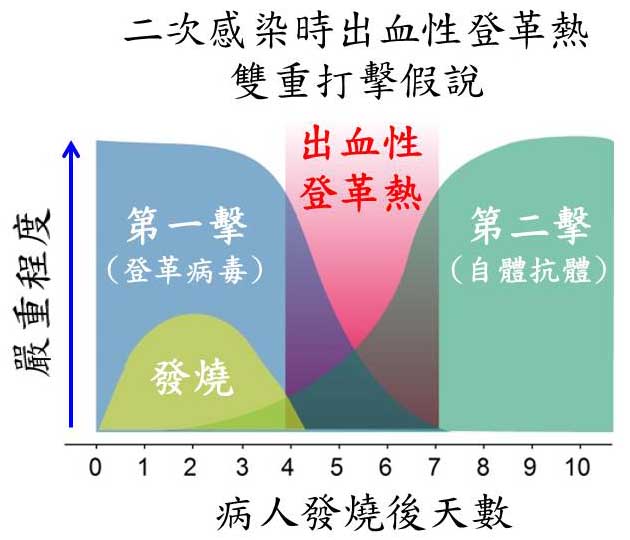
慈濟大學生科院,分子生物暨人類遺傳學系的張新侯老師、孫德珊老師以及醫學院,醫學檢驗生物技術學系的吳文陞老師團隊,為出血性登革熱提出了雙重打擊假說。其理論基礎如下: 在第一次感染時登革病毒本身造成之病理反應為”第一打擊”,但並不足以導致出血及死亡病變,但是在二次感染時,病人體內可進一步誘發出自體反應性抗體之”第二打擊”,此時在”第一擊”及”第二擊”雙重打擊之下,即可誘發出血及死亡病變。運用這個假說,慈大研究團隊成功運用登革病毒,以及自體反應性抗體在小鼠模型模擬登革出血性病變。應用此小鼠模型,今年分別在”血栓與凝血” (Thrombosis and Haemostasis)以及”免疫學期刊”(Journal of Immunology)兩知名國際期刊發表論文1,2,並於十月份於第十屆亞太醫用病毒國際會議中,以此雙重打擊假說發表演說3,得到與會專家學者的諸多回響。
張新侯老師表示,雙重打擊假說如果成立,相關自體反應性抗體偵檢試劑的開發 以及檢測,可在病人體內自體反應性抗體尚未到達發病閾值之前予以偵測,達到早期偵測早期治療的效果。此外,相關成果也有可能應用在新藥的篩選。由於登革病毒非結構NS1抗原可能與誘發自體抗體有關。如何避免自體反應性抗體被誘導,是未來登革疫苗之開發中的重要議題。
論文引用References:
- Lien TS, Sun DS, Chang CM, Wu CY, Dai MS, Chan H, Wu WS, Su SH, Lin YY, Chang HH. Dengue virus and antiplatelet autoantibodies synergistically induce haemorrhage through Nlrp3-inflammasome and FcγRIII. Thrombosis and Haemostasis 113, 1060-70 (2015).
- Sun DS, Chang YC, Lien TS, King CC, Shih YL, Huang HS, Wang TY, Li CR, Lee CC, Hsu PN, Chang HH. Endothelial Cell Sensitization by Death Receptor Fractions of an Anti-Dengue Nonstructural Protein 1 Antibody Induced Plasma Leakage, Coagulopathy, and Mortality in Mice. Journal of Immunology 195, 2743-53 (2015).
- Chang HH. Dengue Virus and Antiplatelet Autoantibodies Synergistically Induce Hemorrhage in the Two-hit Mouse Model. 10th Asia-Pacific Congress of Medical Virology (APCMV), OS 4-4, Oct/17 (2015).
附圖: 出血性登革熱雙重打擊假說,以參考文獻中之模型所繪製1,2。